Tobacco business
Over the last years, the main international tobacco players expanded their portfolio in both combustibles*1 and Reduced-Risk Products (RRP)*2 to offer more choice to consumers. JT Group’s tobacco business offers products to consumers across all combustibles and RRP segments.
We expect the global combustibles industry value to continue growing in the foreseeable future, while the global RRP industry is expected to grow both in terms of volume and value. Among RRP segments, we expect heated tobacco sticks (HTS)*3 to be the largest growth driver, followed by e-vapor and modern oral. The growth rate will ultimately depend on innovation, consumer acceptance, regulation and taxation of RRP.
- *1 Combustibles: Combustibles include all tobacco products excluding contract-manufactured products and RRP
- *2 Reduced-Risk Products (RRP): Products with the potential to reduce the risks associated with smoking. In JT’s portfolio, these products include heated tobacco products (HTP), e-vapor and oral
- *3 Heated tobacco sticks (HTS): High temperature heated tobacco products. One stick is equivalent to a stick of cigarettes
Notes:
- The word consumers used in the context of the tobacco business means adult consumers. Minimum legal age for smoking varies in accordance with the legislation in each country
- This section is intended only to explain the business operations of the JT Group, not to promote sales of tobacco or nicotine containing products or encourage smoking or using nicotine containing products
For more details on our approach to responsible marketing, please refer to Our marketing standards.
In the following four strategic focus areas, we cover our approach to sustainability across our tobacco business.
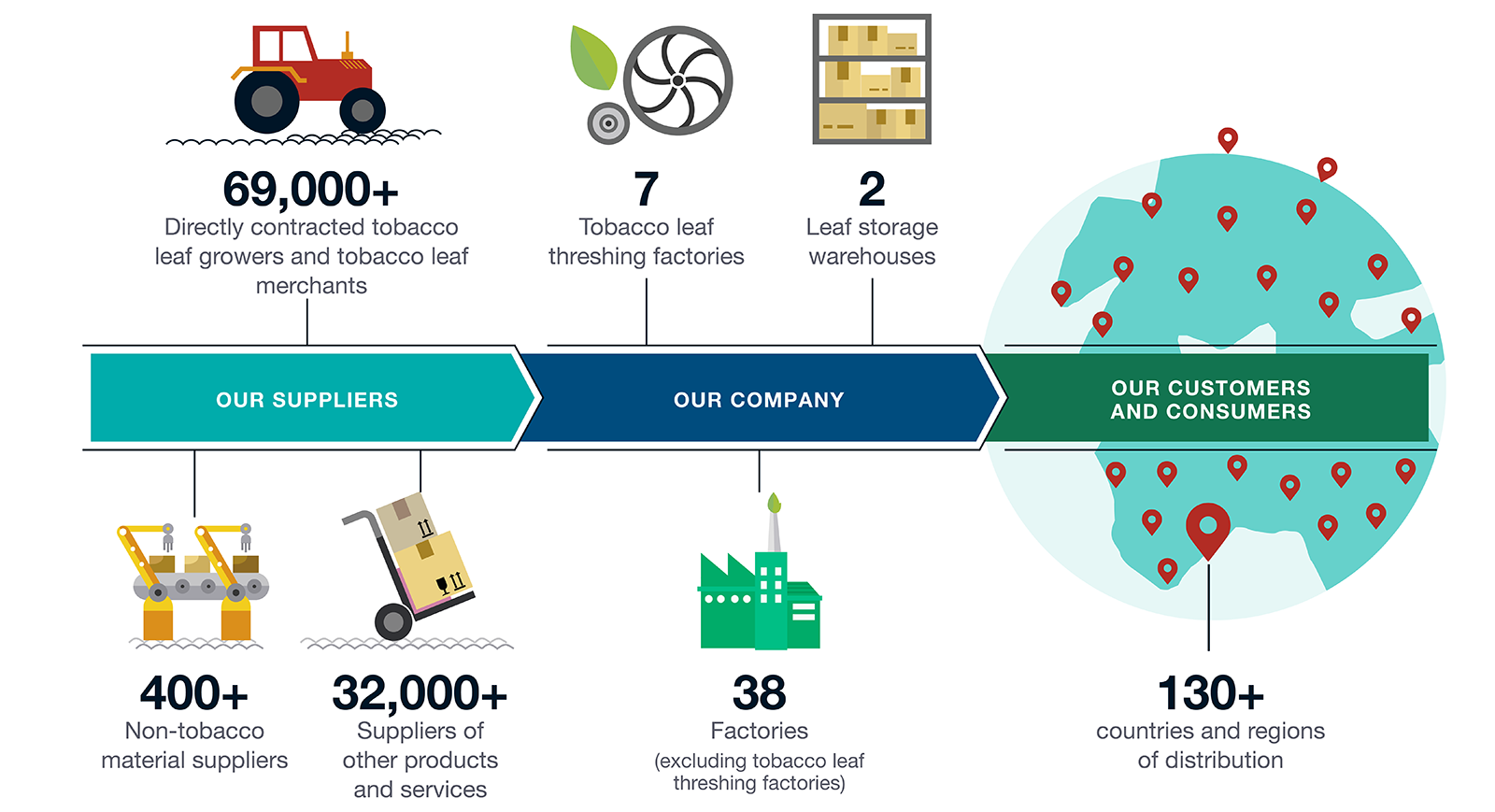
Our tobacco business value chain